Water compliance testing for medical and allied health practices
Any practice that reprocesses reusable instruments — podiatry, GP, physiotherapy, specialist clinics and day surgery — has one water testing obligation under AS 5369:2023. We handle it — monthly routine monitoring and annual audit — with NATA-accredited results and zero admin on your end.

Your Compliance Obligations
Products and Pricing
Everything your practice needs. Sample kit, courier and accredited report Included.
ex GST · Year 1
AS 5369:2023 requires monthly testing of the water in your sterilisation room. Routine monitoring confirms bacteria levels and key chemistry parameters are within specification every month.
per annual submission
Once a year, AS 5369:2023 requires a full parameter screen of your washer-disinfector final rinse water. This goes beyond the monthly routine test to include endotoxins, iron, phosphates and silica.
per annual submission
Your autoclave requires chemically pure feedwater to function correctly and protect instruments. AS 5369:2023 Table 7.4 sets strict chemistry and metals limits for dedicated steam generator feedwater.
per annual submission · saves $279 vs separate
If your washer-disinfector and autoclave are fed from the same RO water supply, a single sample satisfies both Table 7.2 and Table 7.4 in one submission. The most common scenario for practices with a single RO unit — and the most cost-effective option. Order now → Kit dispatched within one business day of order.
How It Works
-

Step 1 - We send you a kit
A sample kit is sent to your business. Everything you need is included.
-

Step 2 - Collect the sample
The process takes about 5 minutes. Step-by-step instructions included. Your practice manager or dental assistant can do this.
-

Step 3 - Priority pick up
Call the priority number in your kit, quote your pre-paid barcode, and a courier collects directly from your reception desk — no accounts, no paperwork, no cost to you.
-
Step 4 - Receive your results
Your NATA Certificate of Analysis and audit-ready compliance report are emailed within 5–7 business days — benchmarked against AS 5369:2023 limits and ready to file for your next accreditation review.
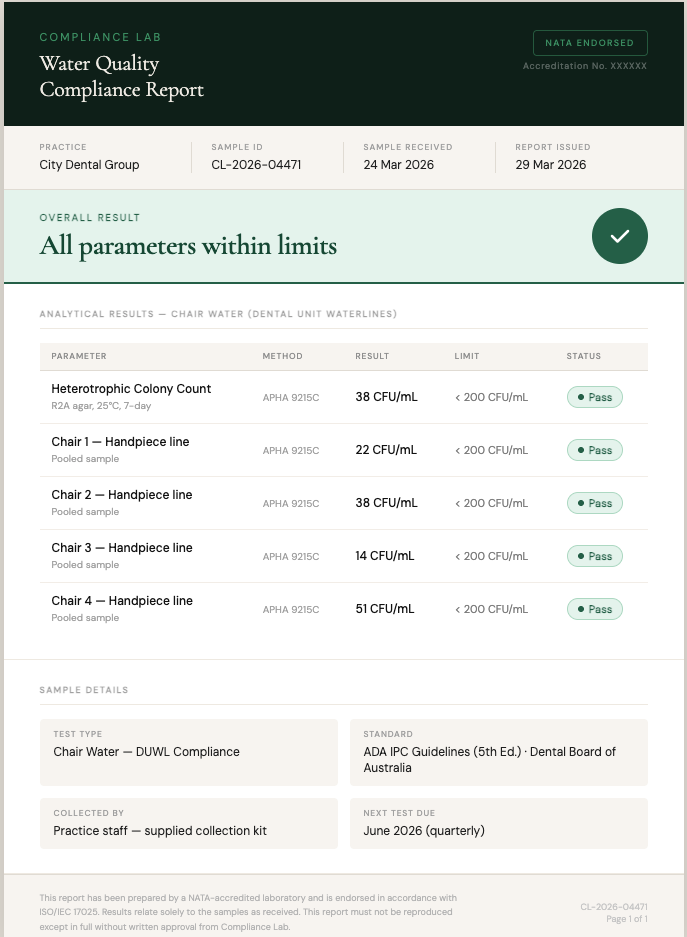
Your compliance report, clearly explained
Every test includes a NATA-endorsed Certificate of Analysis and an audit-ready compliance report. Your report shows each parameter, the result, the regulatory limit, and whether it is within specification. Everything you need to file for your compliance records.
Issued by a NATA-accredited laboratory under ISO/IEC 17025
Results benchmarked against AS 5369:2023 limits
NATA accreditation under ISO/IEC 17025 is the recognised standard for documentary evidence in any professional board or accreditation body review
Emailed within 5–7 business days of sample receipt
NATA Certificate of Analysis attached as PDF — ready to file
Example report uses fictitious practice and sample data.Compliance Report
| Parameter | Result | Limit | Status |
|---|---|---|---|
| pH | 6.4 | 5.0–7.0 | Within limits |
| Conductivity | 18 µS/cm | ≤30 µS/cm | Within limits |
| Total Hardness | <1 mg/L | ≤2 mg/L | Within limits |
| Chloride | 8.2 mg/L | ≤50 mg/L | Within limits |
| Reactive Phosphate | 0.08 mg/L | ≤0.3 mg/L | Within limits |
| Silica (Reactive) | 0.12 mg/L | ≤1.0 mg/L | Within limits |
| Iron (total) | <0.02 mg/L | ≤0.2 mg/L | Within limits |
| Parameter | Result | Limit | Status |
|---|---|---|---|
| Bacterial EndotoxinsLAL kinetic-chromogenic | 0.04 EU/mL | ≤0.25 EU/mL | Within limits |
| Heterotrophic Colony CountR2A agar, membrane filtration | <1 CFU/100mL | ≤100 CFU/100mL | Within limits |
Page 1 of 1